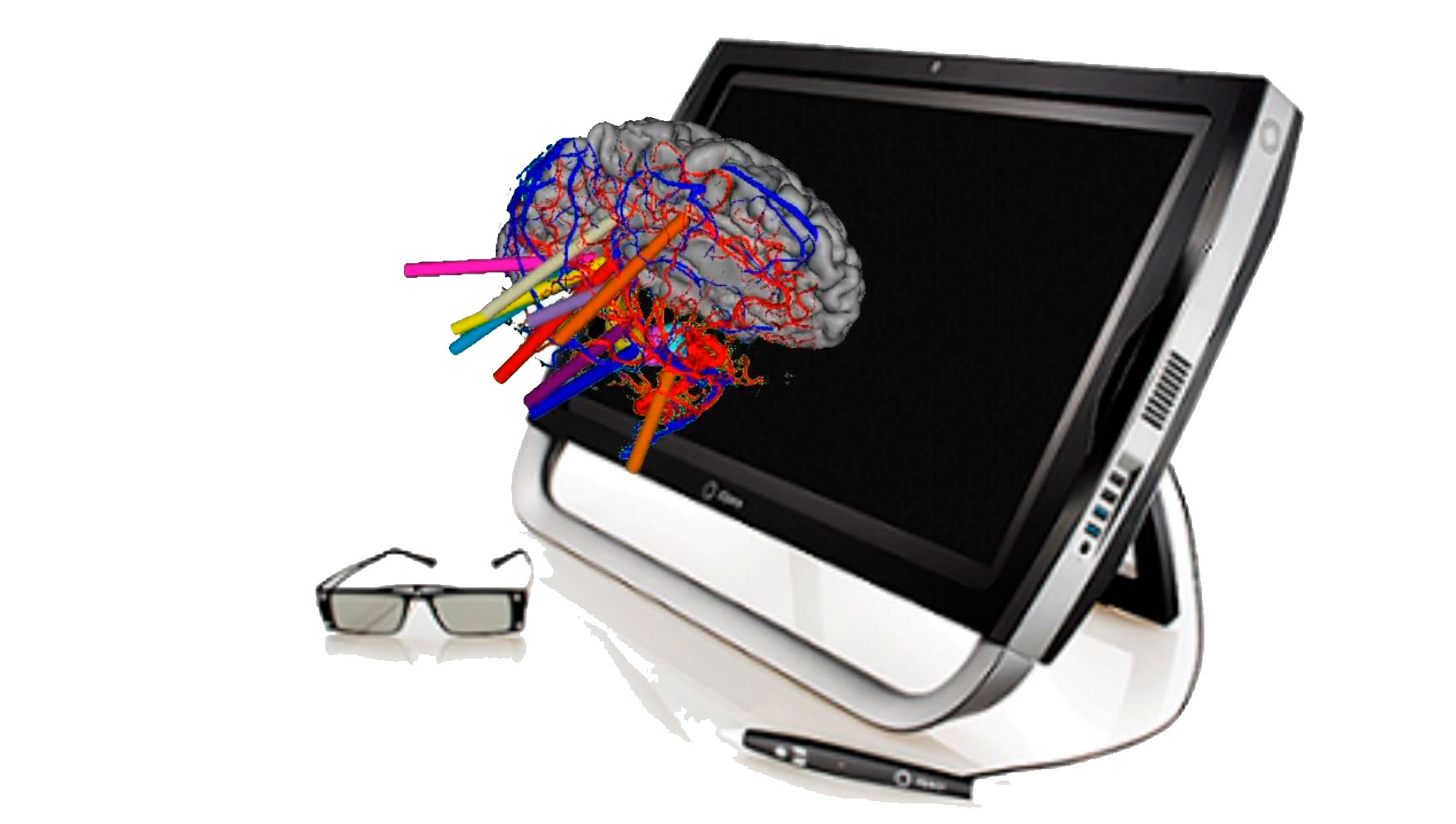
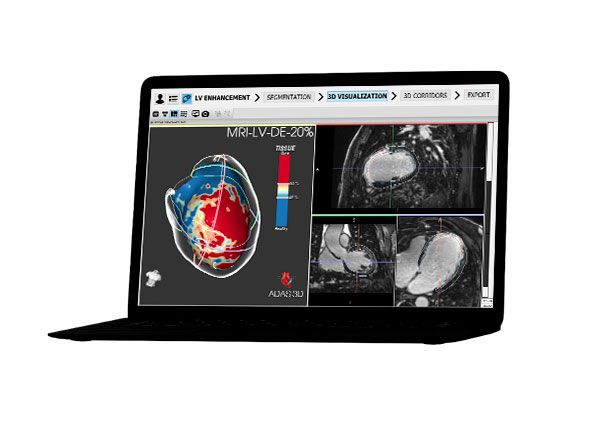
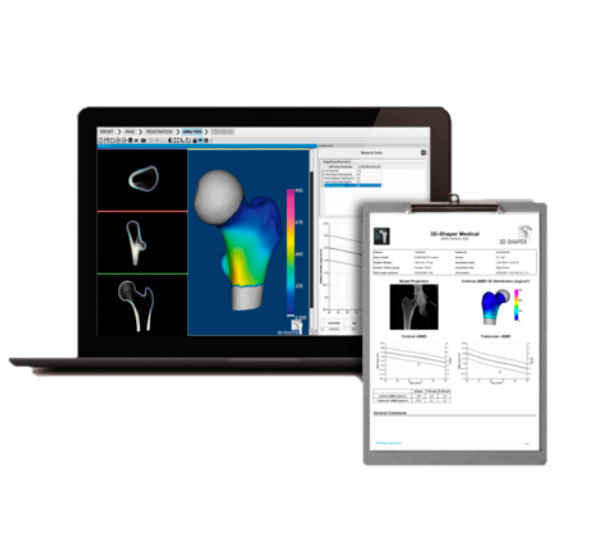
Galgo has focused on four
medical disciplines:
To facilitate the multidisciplinary decision-making and planning of stereotactic neurosurgical interventions.
Investigational Product of Galgo Medical
To help qualified healthcare professionals visualize fibrosis, wall thickness and surrounding anatomical structures.
Spin-off company of Galgo Medical
To improve bone disease diagnostics by turning DXA imaging into 3D patient specific models.
Spin-off company of Galgo Medical
About us

Galgo Medical SL is a Barcelona-based company incorporated in July 2013. It emerged from the Department of Information Technologies (DTIC) of the Universitat Pompeu Fabra (Barcelona, Spain).
The team developed a solid experience in developing imaging post-processing software solutions obtained during more than seven years working together at the Universitat Pompeu Fabra. They were involved in national and international competitive projects and nurtured over the years close collaborations with several clinical institutions.
Key Personnel
In the picture, from left to right:
- Héctor Fernández, co-founder, ex-CTO of ANKYRAS, now with MENTICE AB
- Yves Martelli, co-founder, no longer in GALGO
- Jacobo Gil de Biedma, CRO of GALGO Medical SL and Adas3D Medical SL
- Luis Serra (PhD), co-founder, President of the Board of GALGO Medical SL and Adas3D Medical SL
- Ludovic Humbert (PhD), co-founder, CEO of 3D-SHAPER Medical SL
- Martin Steghöfer, Principal engineer, Adas3D Medical SL
- Xavi Planes, co-founder, CTO of Adas3D Medical SL
- Antoni Riu, co-founder, CEO of GALGO Medical SL and Adas3D Medical SL
(Not appearing, alphabetical order)
- Ignacio Delgado (MD PhD), CSO of StereoDive
- Rosa Maria Figueras (PhD), CCO of Adas3D Medical SL
- Francia Rojas, Quality and Regulatory Manager of GALGO Medical SL, Adas3D Medical SL and 3D-SHAPER Medical SL.
- Alfredo Higueras (PhD), Director of StereoDive
- María Izquierdo, CTO of 3D-SHAPER Medical SL
- Ignacio Larrabide (PhD), co-founder, ex-CCO of ANKYRAS, now with CONICET and MENTICE AB
- Sam Wilson, CRO of 3D-SHAPER Medical SL
Funded Projects
Galgo has participated in the following projects:
-
2021

Ministerio de Ciencia e Innovación - PLEC2021-007614 "DIDIMO" - Personalized cardiac digital twins to enhance the development of medical devices and cardiac antiarrhythmic treatments. -
2020

Horizon2020 - H2020-MSCA-ITN-2020 – #955735 “DISc4ALL” – European Training Network to advance integrated computational simulations in translational medicine applied to intervertebral disc degeneration.Eurostarts E!114276 “ANKLOUD” – financiación CDTI CIIP-20202012 – Simulation- based platform for personalized brain aneurysm treatment with braided devices.
-
2019

RETOS 2019-007413-1 “ANDAMIO” – Estimation of the mechanical resistance of the spine to prevent osteoporotic fracturesCDTI – Proyectos unilaterales – IDI-20191246 “INTOPHARMA” – Development of a new tool for personalization in the pharmacological treatment of osteoporosis andits subsequent follow-up.
-
2018

CDTI PID IDI-20180908 “VISOR” - Multimodal 3D viewer for planning and navigating cardiac ablation procedures -
2017

ICEX NEXT – Fondo Europeo de Desarrollo Regional – Commercial development plan for the product 3D-SHAPER -
2016

MINECO RTC-2016-5073-1 POROVIEW – Predictive modeling of braided devices (flow-diverters)CDTI INNO-20161015 INNOGLOBAL VETA – Risk stratification of sudden cardiac death in patients with non-ischemic cardiomyopathy
TORRES QUEVEDO PTQ-16-08627 – Dr PhD Renaud Winzenrieth
TORRES QUEVEDO PTQ-16-08628 – Dr MD PhD Ignacio Delgado
ENISA Crédito 2016 para el Desarrollo Empresarial de nuevas empresas de base tecnológica (Credit for the Business Development in new technology companies)
-
2015

MINECO RTC-2015-3515-1 ADAS – Heart modeling for planning cardiac ablation proceduresCDTI SNEO-20151062 NEOTEC PPMED – Programa de impulso a nuevas empresas (Support program for new companies)
TORRES QUEVEDO PTQ 15-07782 – Dra Phd Rosa Maria Figueras
-
2014

CDTI IDI-20150015 IBEROEKA FDF-MOBILE Co-financed by FONDO EUROPEO DE DESARROLLO REGIONAL (FEDER), within the program Programa Operativo de Crecimiento Inteligente 2014-2020 – Braided Device Modeling for Brain Aneurysm Intervention PlanningCDTI CIIP-20141008 EUROSTARS 3D-DXA SPINE – 3D reconstruction of the spine in shape and density from a DXA image
MINECO RTC-2014-2740-1 BIODXA – Biomechanical modeling of the femur for bone densitometry
ENISA Crédito 2014 para el Desarrollo Empresarial de nuevas empresas de base tecnológica (Credit for the Business Development in new technology companies)
-
2013

TORRES QUEVEDO PTQ 2013-6124 – Dr Luis Serra del Molino
TORRES QUEVEDO PTQ 2013-6213 – Dr PhD Ludovic Humbert
GENERALITAT DE CATALUNYA, DOCTORAT INDUSTRIAL DI-2014, CO- FINANÇAMENT FONS SOCIAL EUROPEU – Dra PhD Mirella López
Supported by